Sometimes teacher needs quick reference for science class, but finding handy materials not always easy. They looking for periodic table printout for class activity, make learning fun. Handy, clear, and detailed table hard to find. They want help students understand elements fast.
Crafted a printable periodic table for ease of learning. It's color-coded for distinguishing between metal, non-metal, and metalloid elements. Handy labels and atomic numbers included for quick reference. Great tool for students to familiarize themselves with elements and their properties.

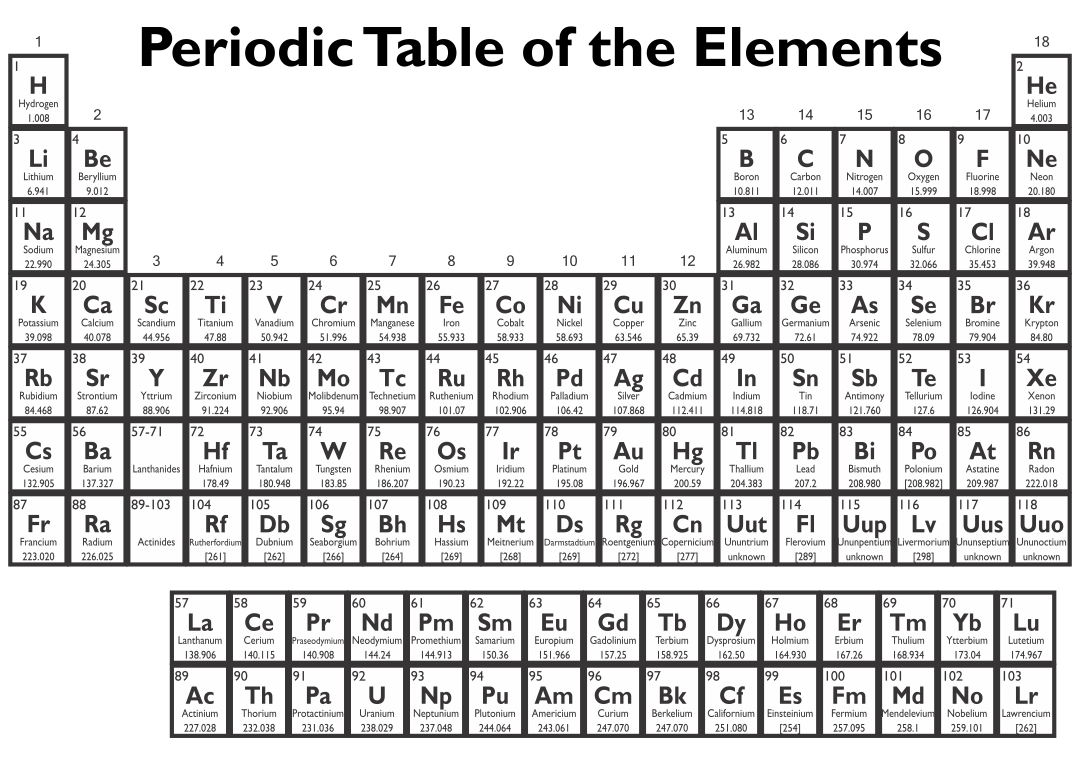
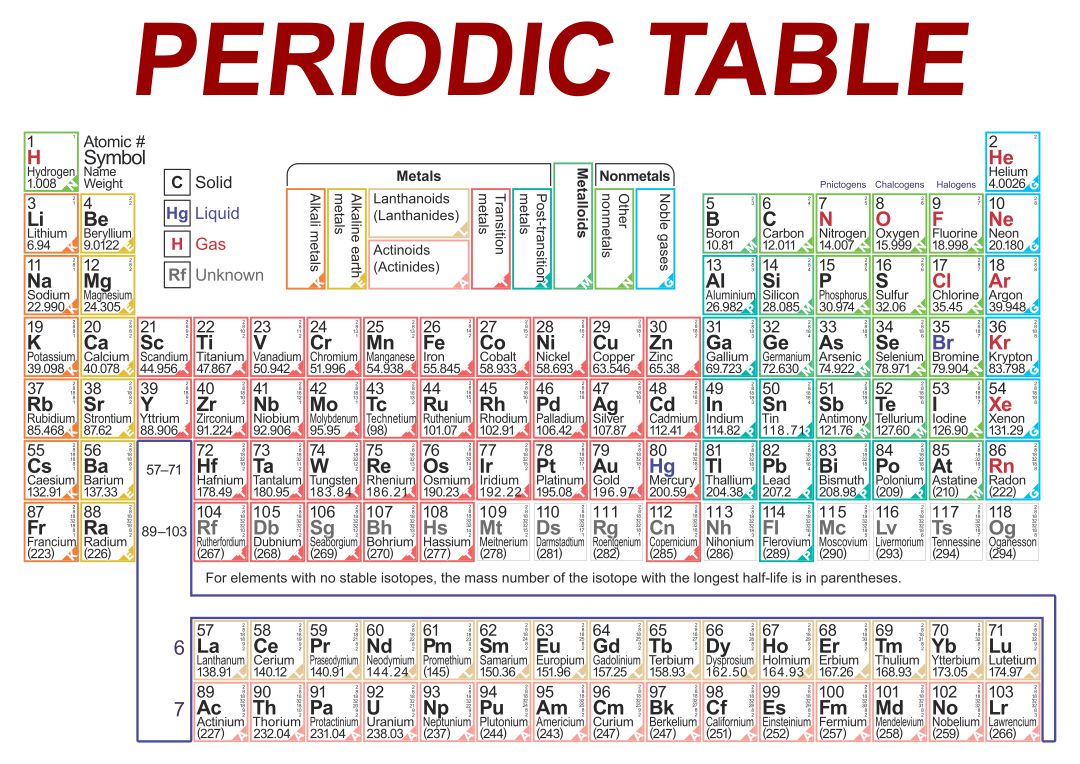
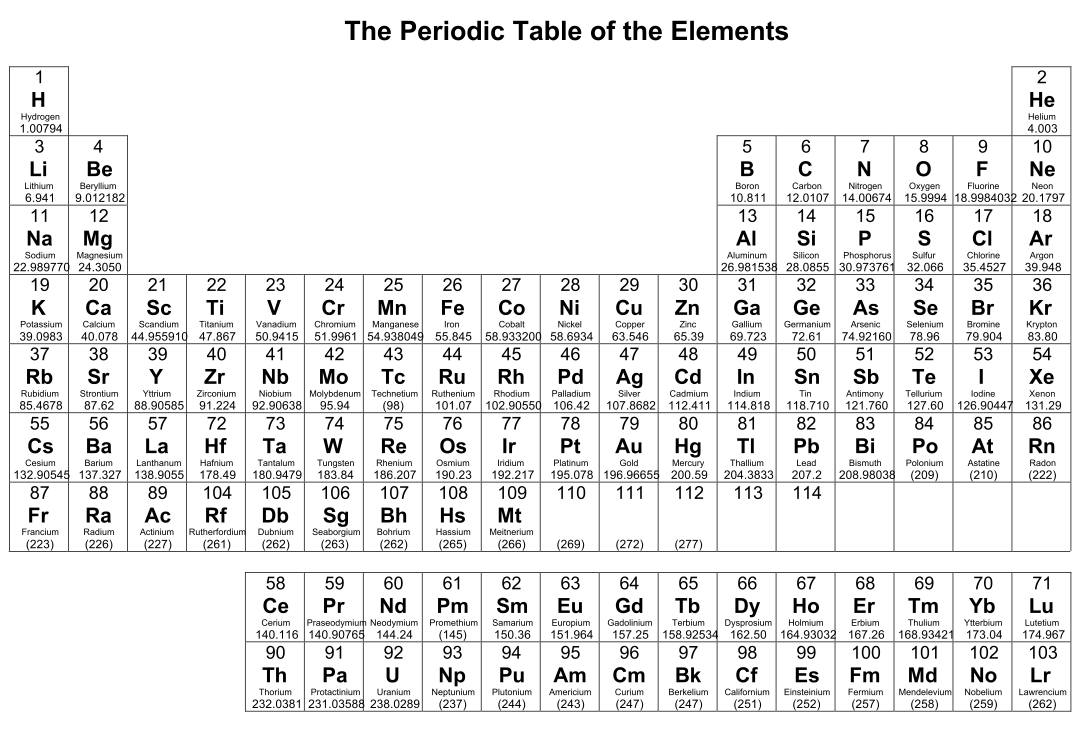
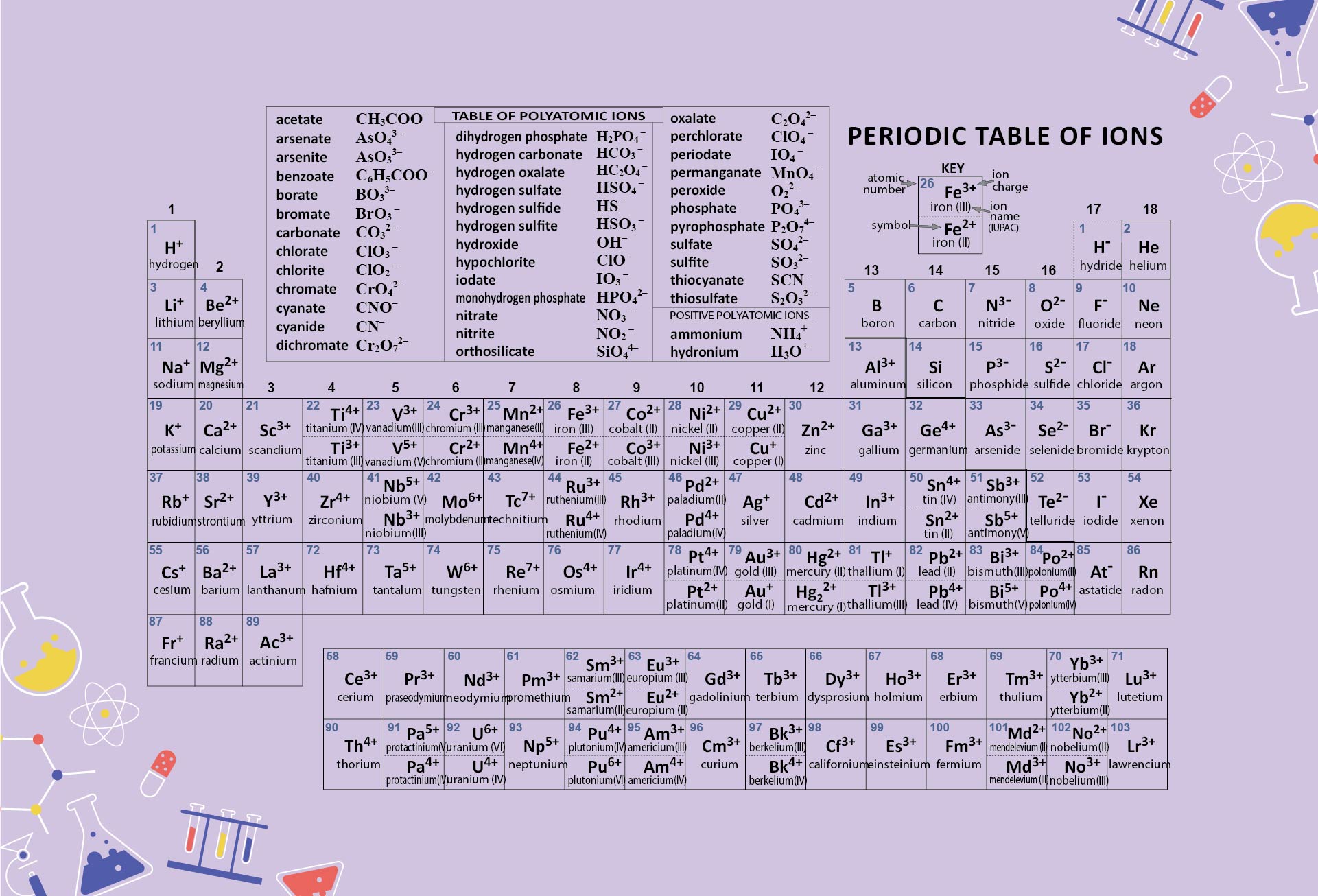
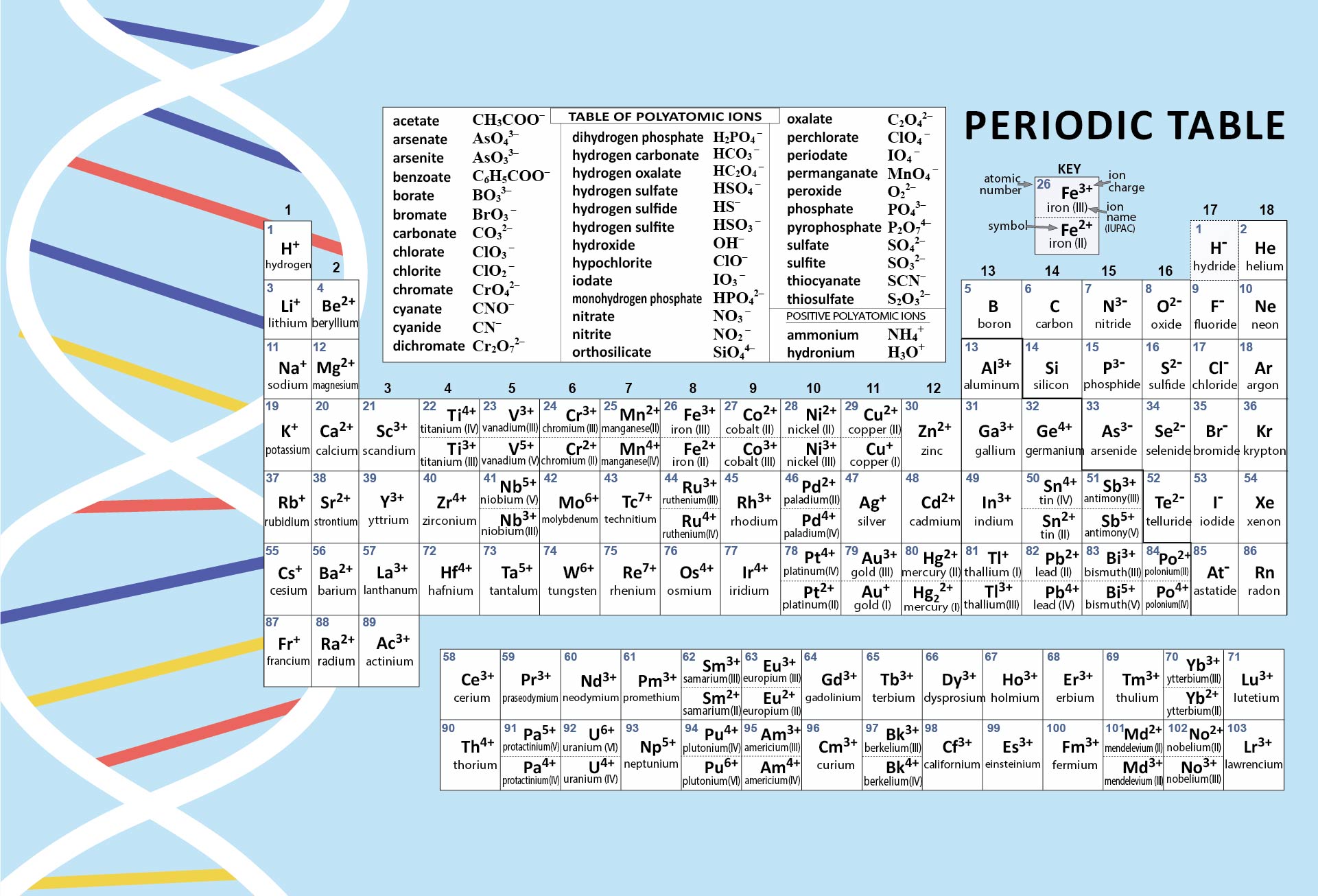
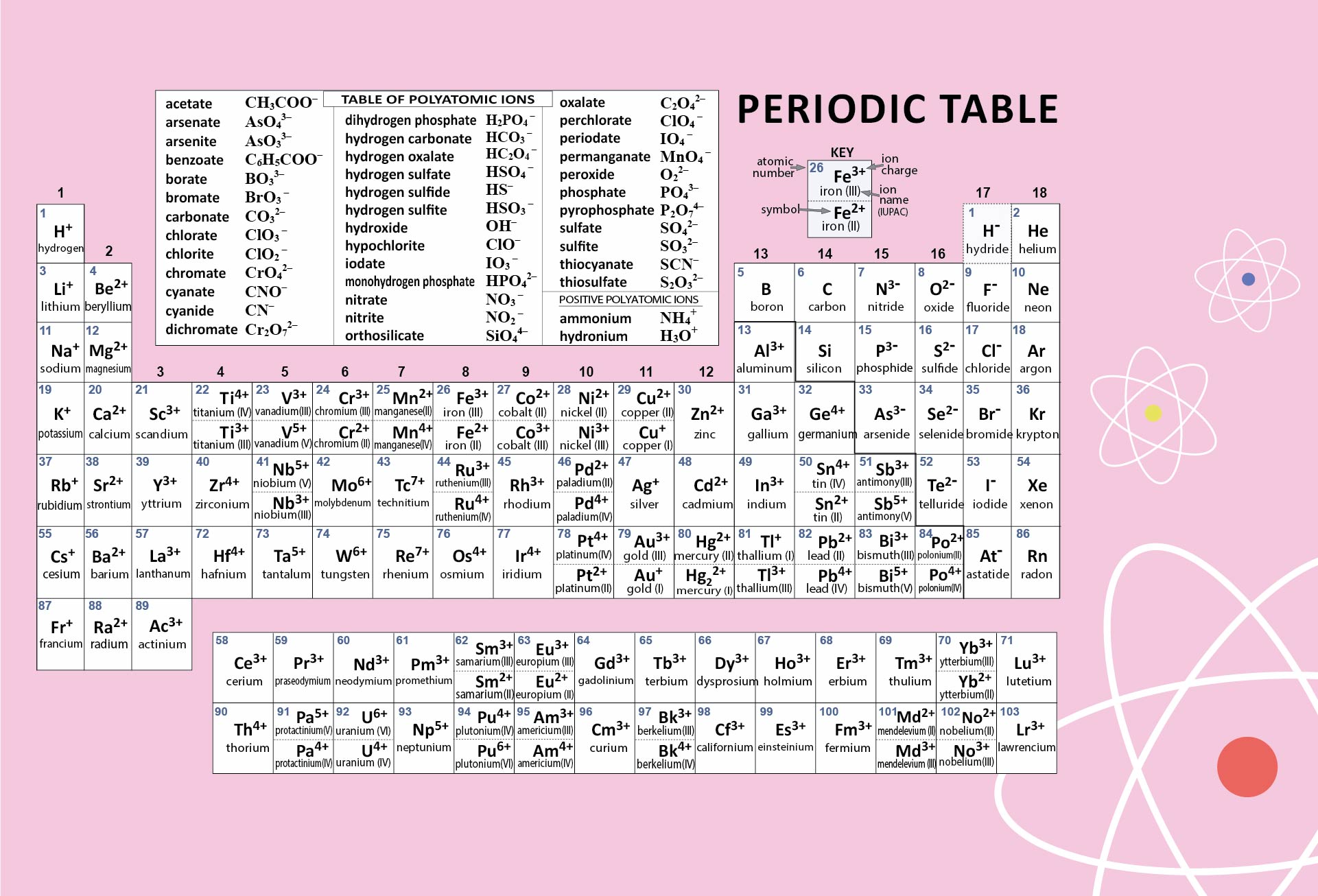
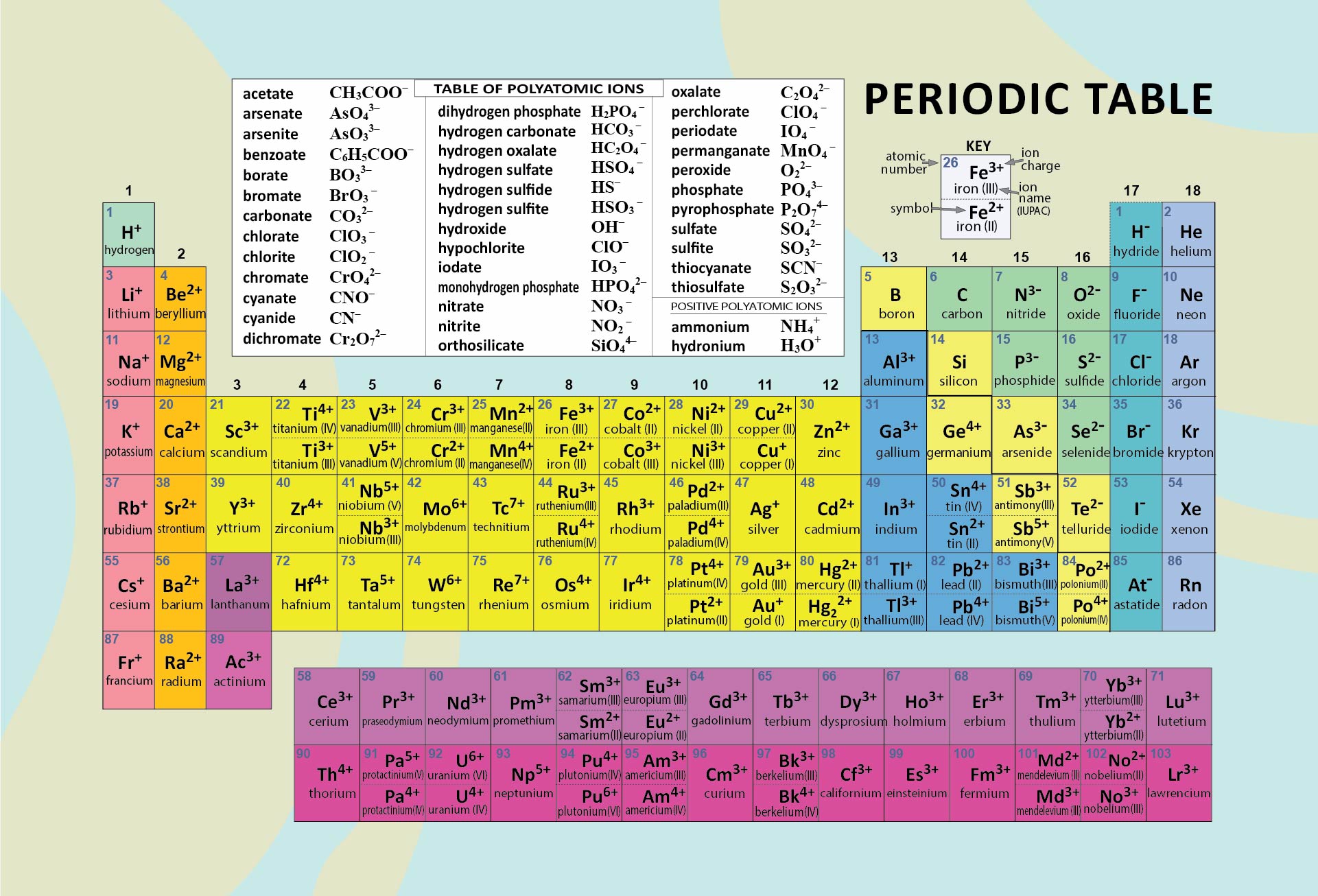
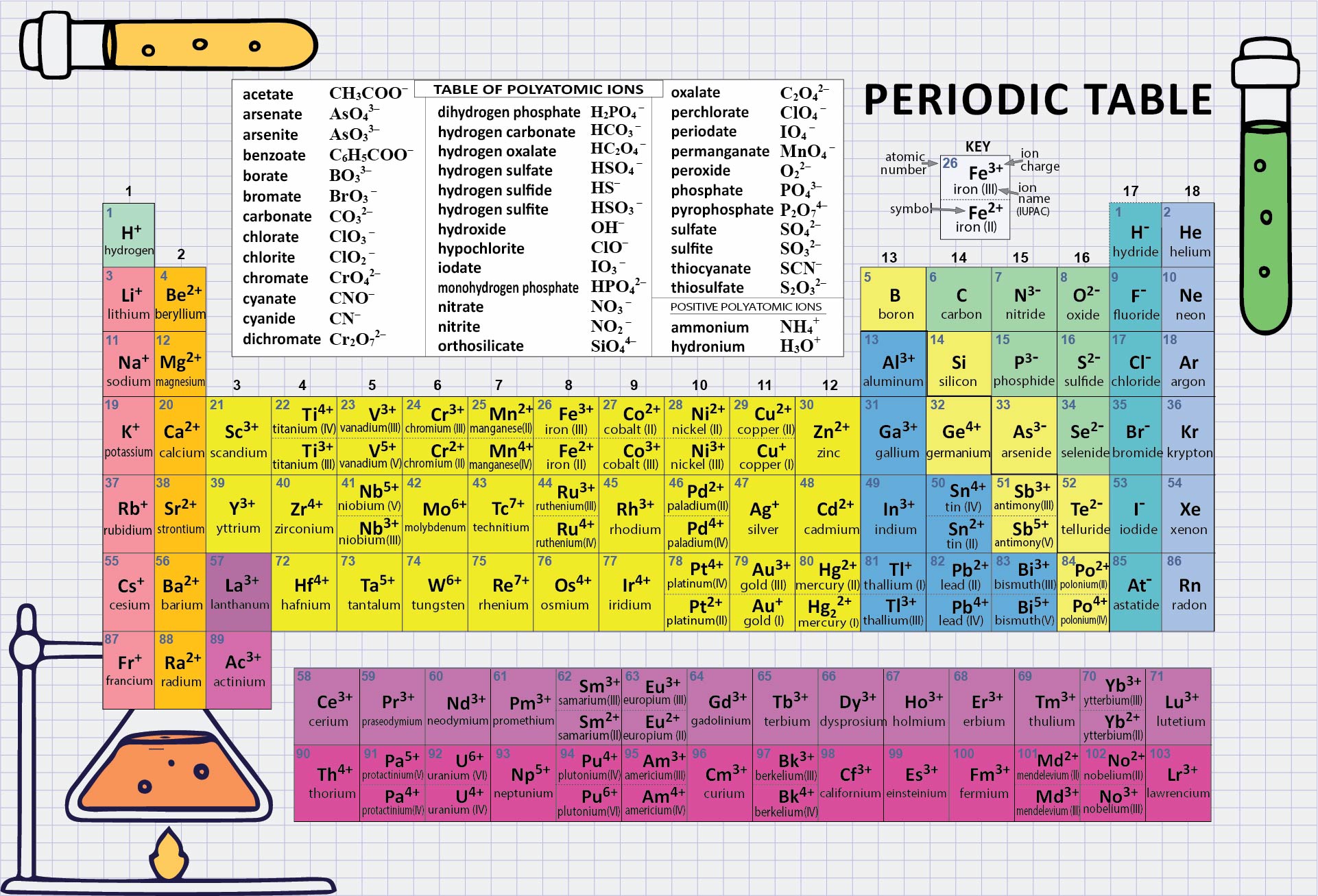


Start by memorizing the order of the seven periods. You can use mnemonics or memory techniques to help you remember the sequence. For example, you could create a phrase like "Happy Little Cats Nap Near Older Rooms" to represent the first letters of each period (1, 2, 3, 4, 5, 6, 7).
The first two periods are relatively simpler to understand as they contain a smaller number of elements. Spend some time familiarizing yourself with these periods before moving on to the others.
Each period exhibits certain periodic trends that can help you understand the arrangement and properties of elements within it. For example, as you move from left to right across a period, atomic number and atomic mass generally increase, and atomic size decreases. Understanding these trends can assist in memorizing the order and properties of elements within a period.
Use a visual representation of the periodic table to aid your learning. Look for periodic tables that highlight the periods and their corresponding elements. Familiarize yourself with the location of each period and the elements within them.
Utilize online resources and interactive tools that allow you to test your knowledge of the periodic table of periods. Many websites and apps offer quizzes and games specifically designed to help you learn and remember the order and properties of elements in each period.
Employ mnemonic techniques or memory aids to associate elements with their corresponding periods. You can create memorable phrases, acronyms, or visual associations to help you recall the order of elements within a period.
Regularly review the periodic table of periods to reinforce your learning. Repeat the order of the periods, practice identifying elements within each period, and test your knowledge periodically to ensure retention.
Have something to tell us?
Recent Comments
I love having this Printable Periodic Table of Elements! It's a handy resource that helps me stay organized and learn about different elements in a straightforward way. Thank you for creating and sharing this!
This printable periodic table is a helpful and concise resource for studying the elements. It's easy to read and will enhance your understanding of chemistry.
I found the Periodic Table of Elements Printable to be incredibly helpful and visually appealing. It enables easy access to essential information, making it a valuable educational resource.