Having a printable copy of the periodic table at your disposal allows you to quickly reference the basic building blocks of chemistry anytime you need.
This convenient tool is perfect for students, educators, and science enthusiasts who want to familiarize themselves with element names, symbols, atomic numbers, and weights without relying on digital devices. It's a handy reference for homework, study sessions, or satisfying your curiosity about the elements that compose the world around you.
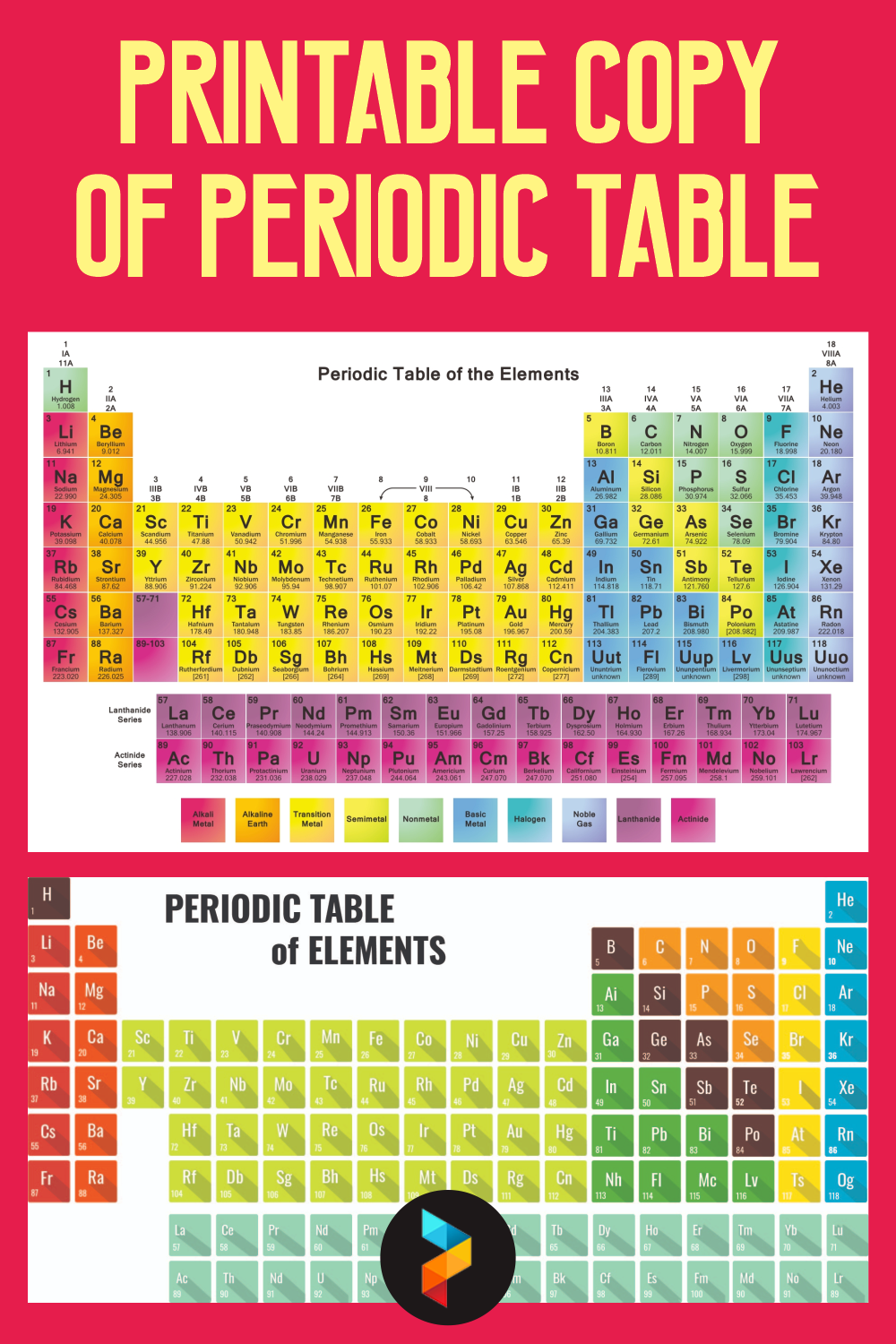
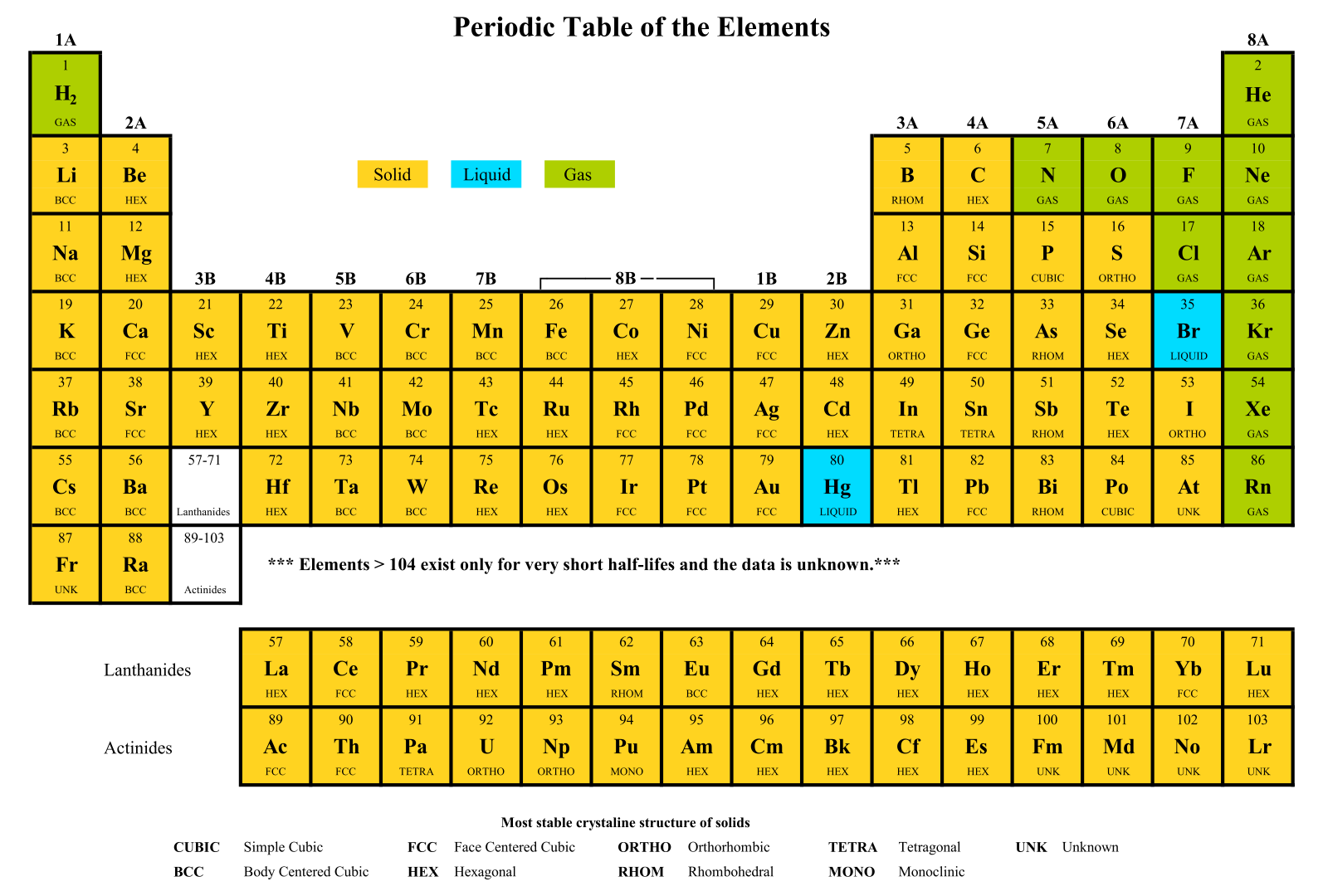
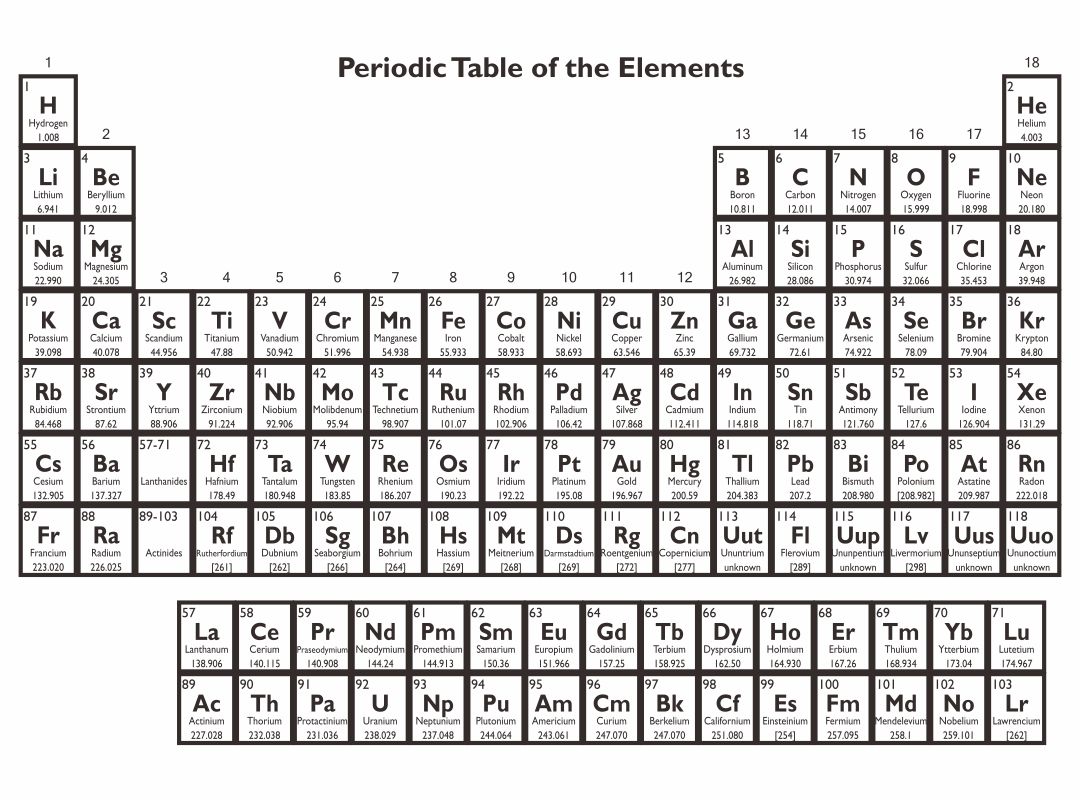

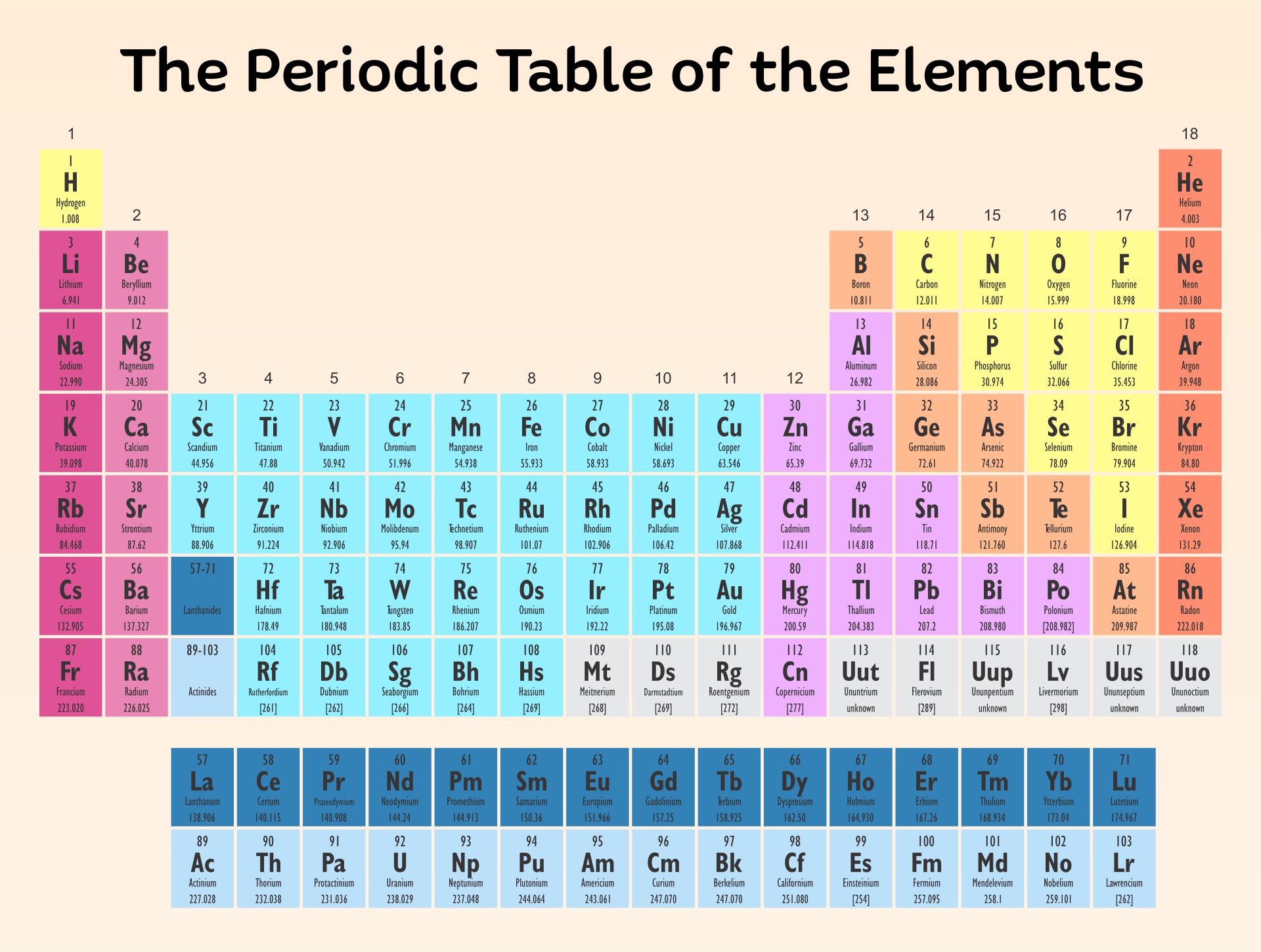
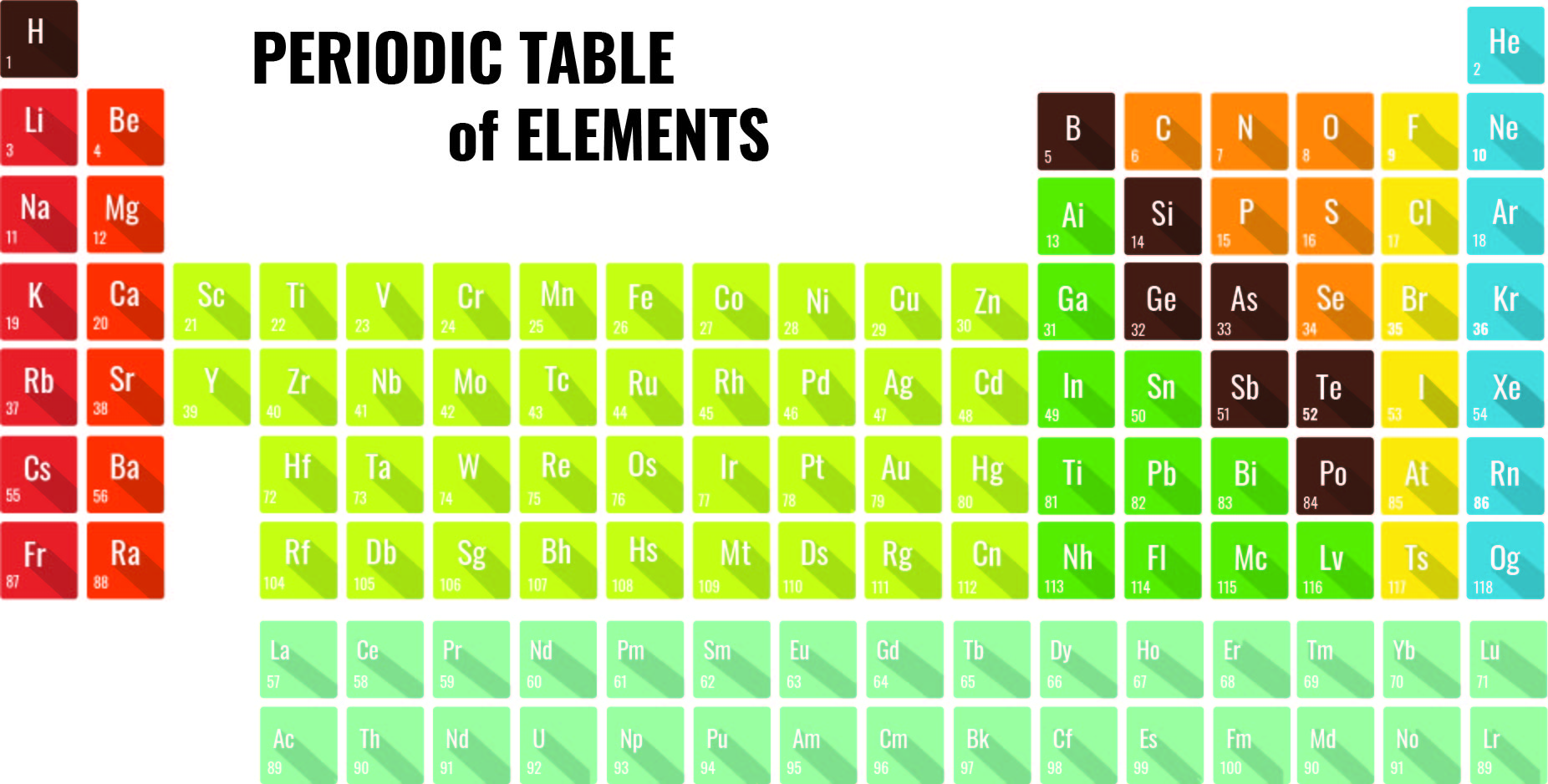
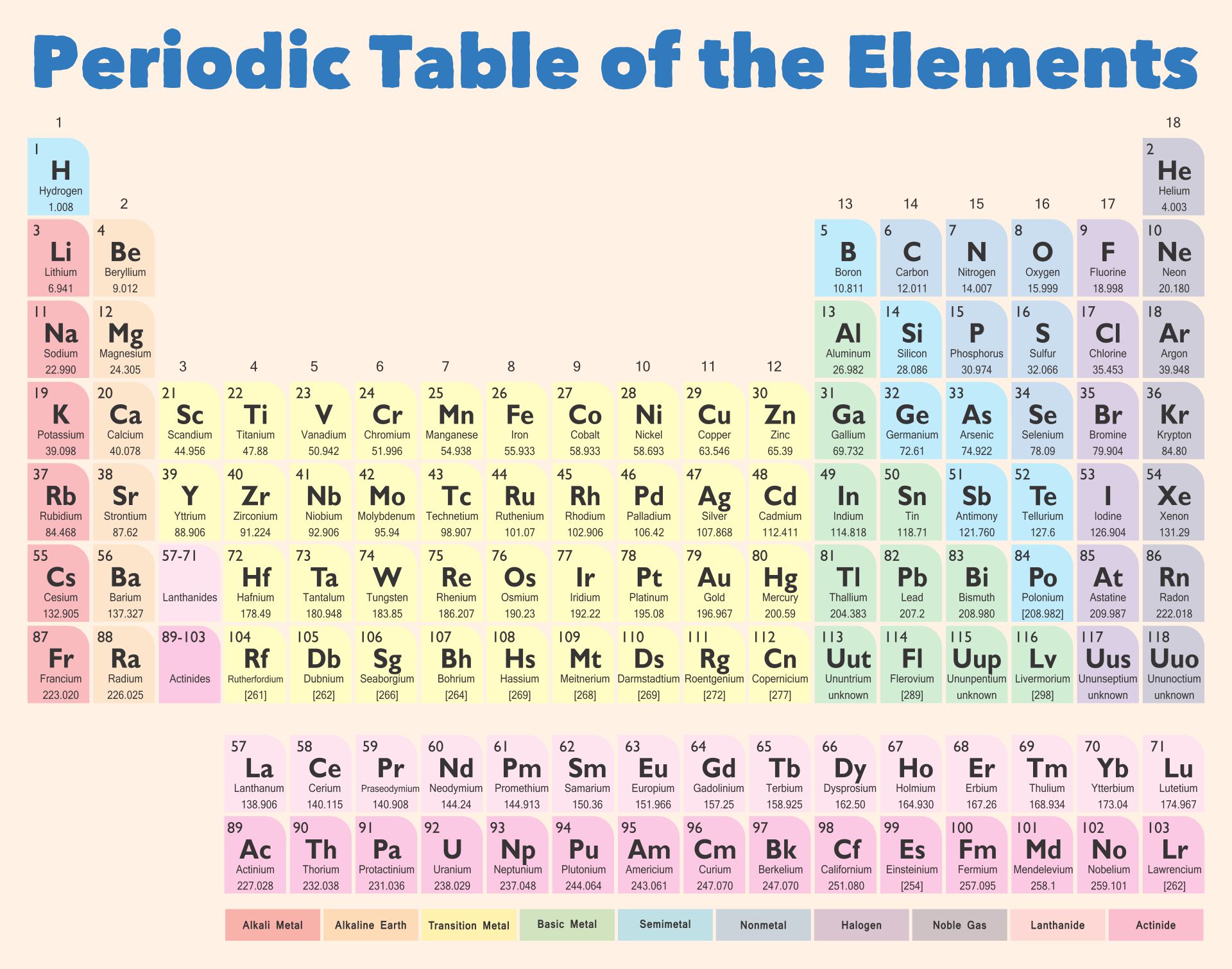
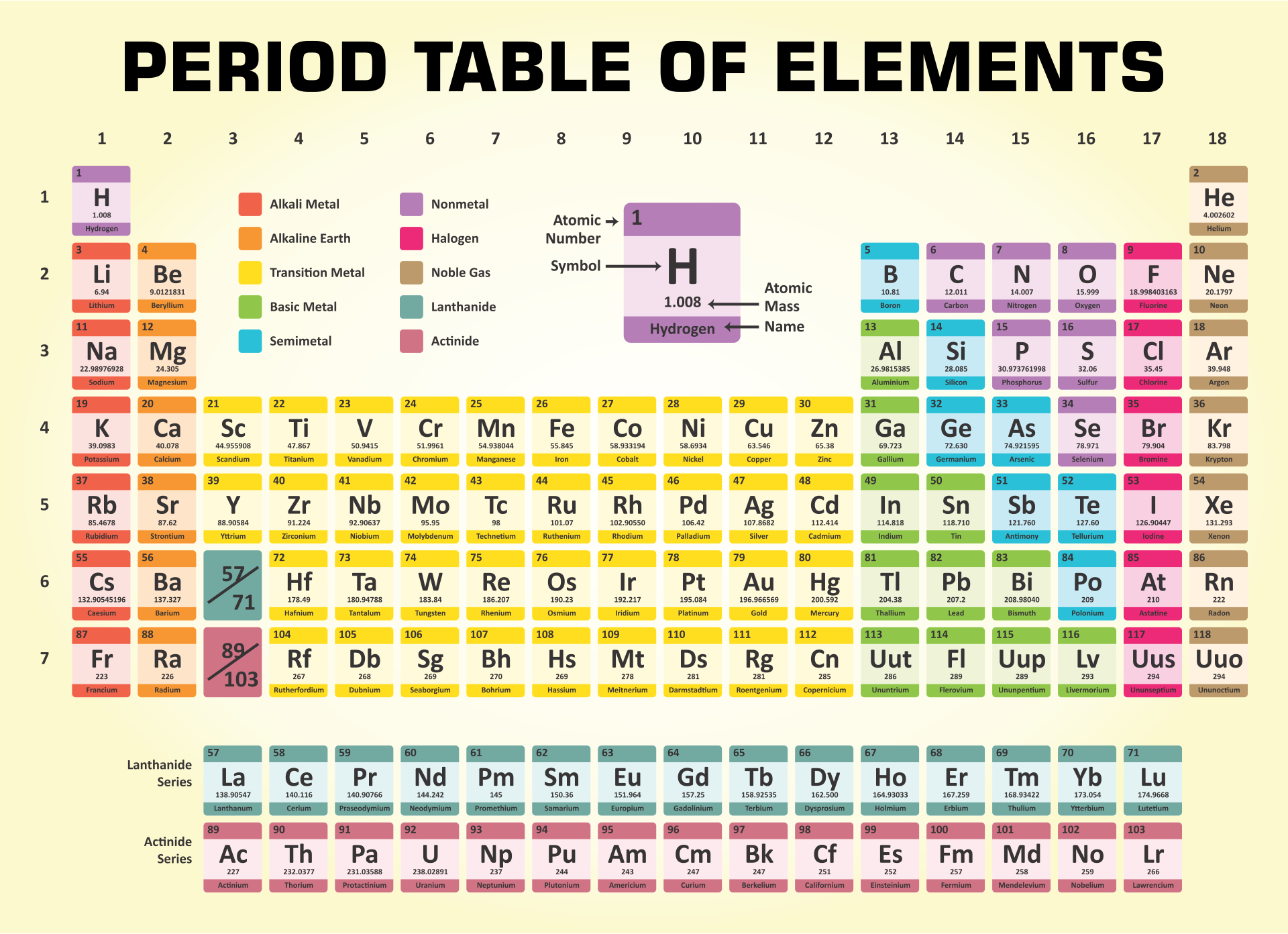
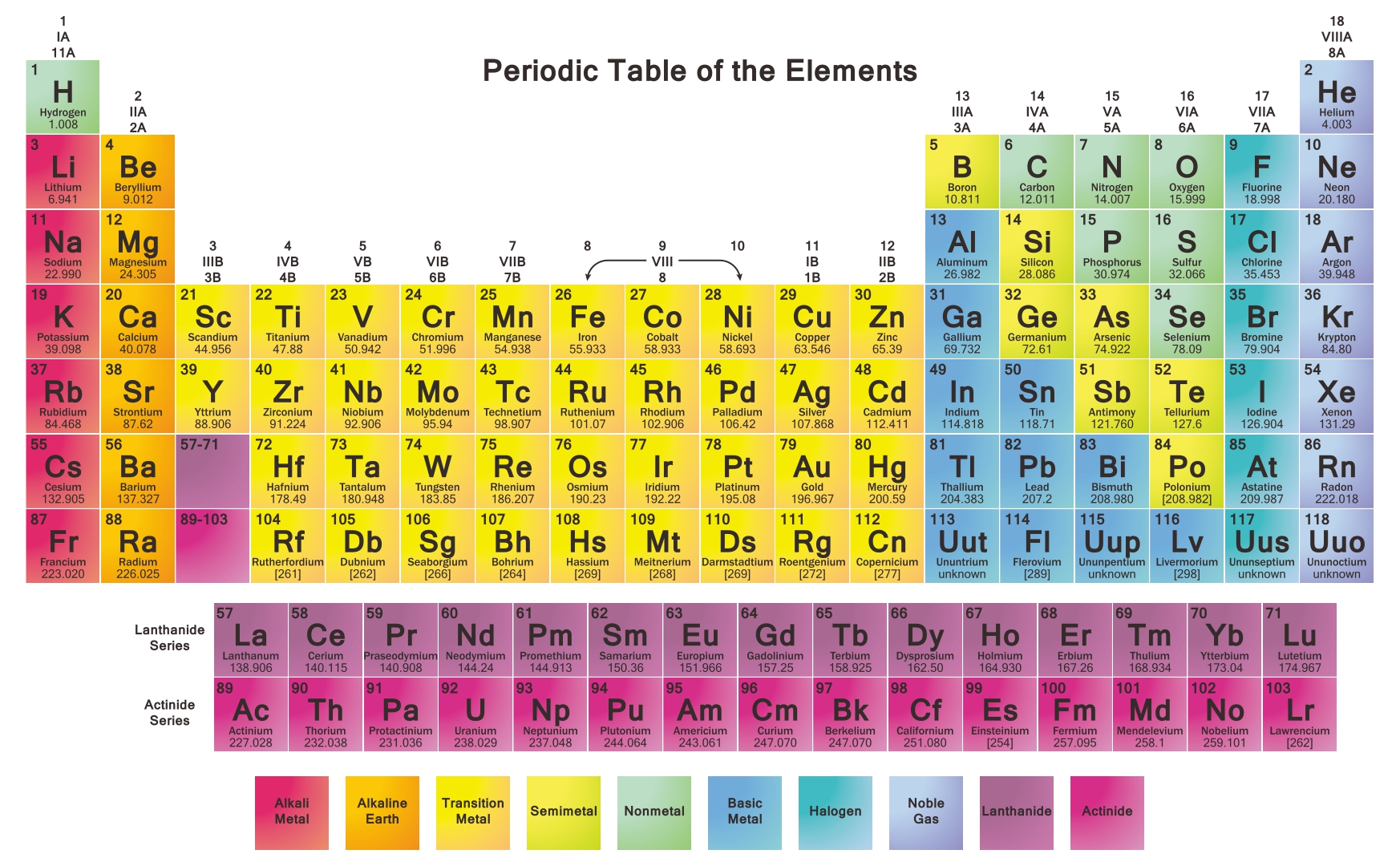
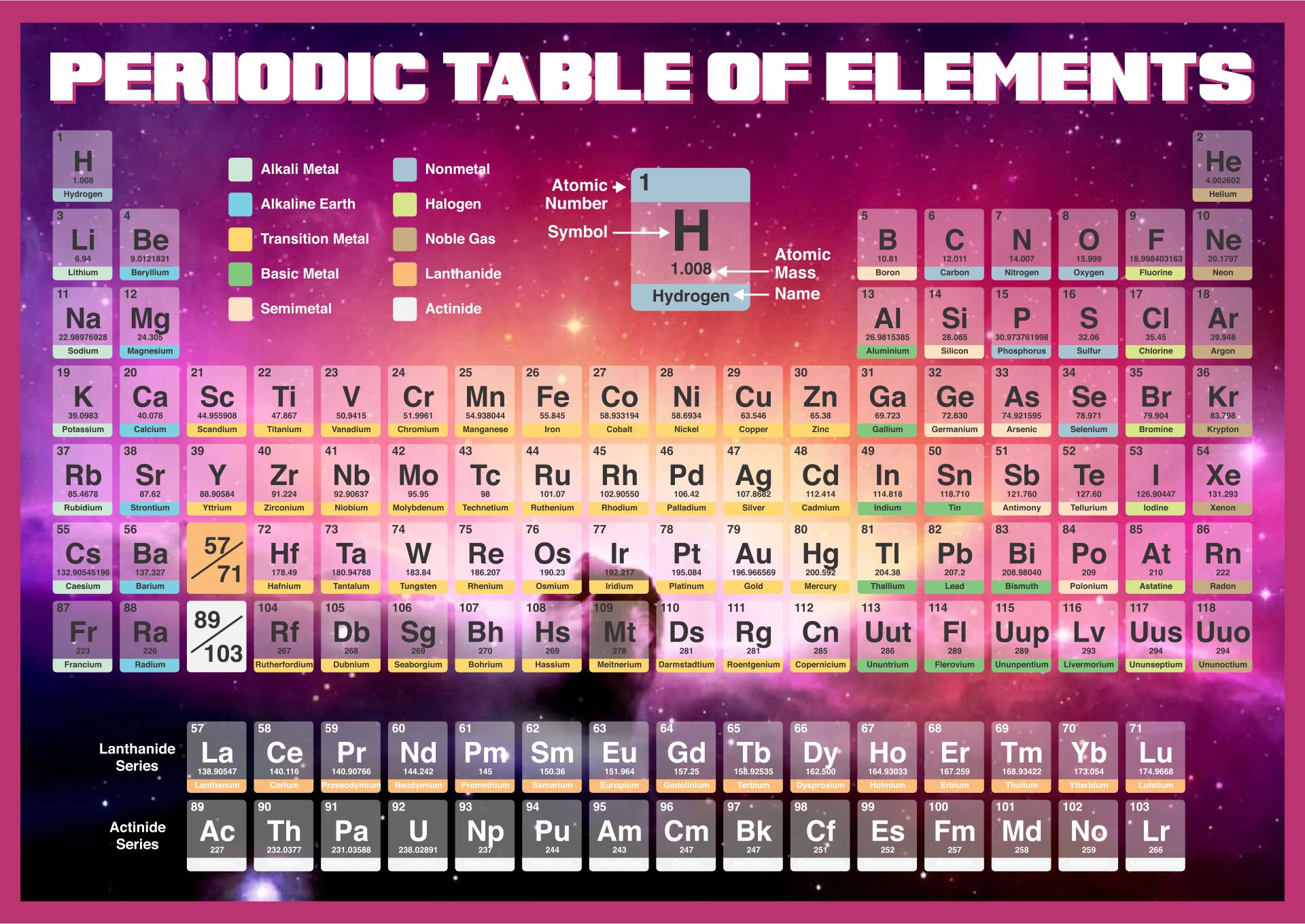
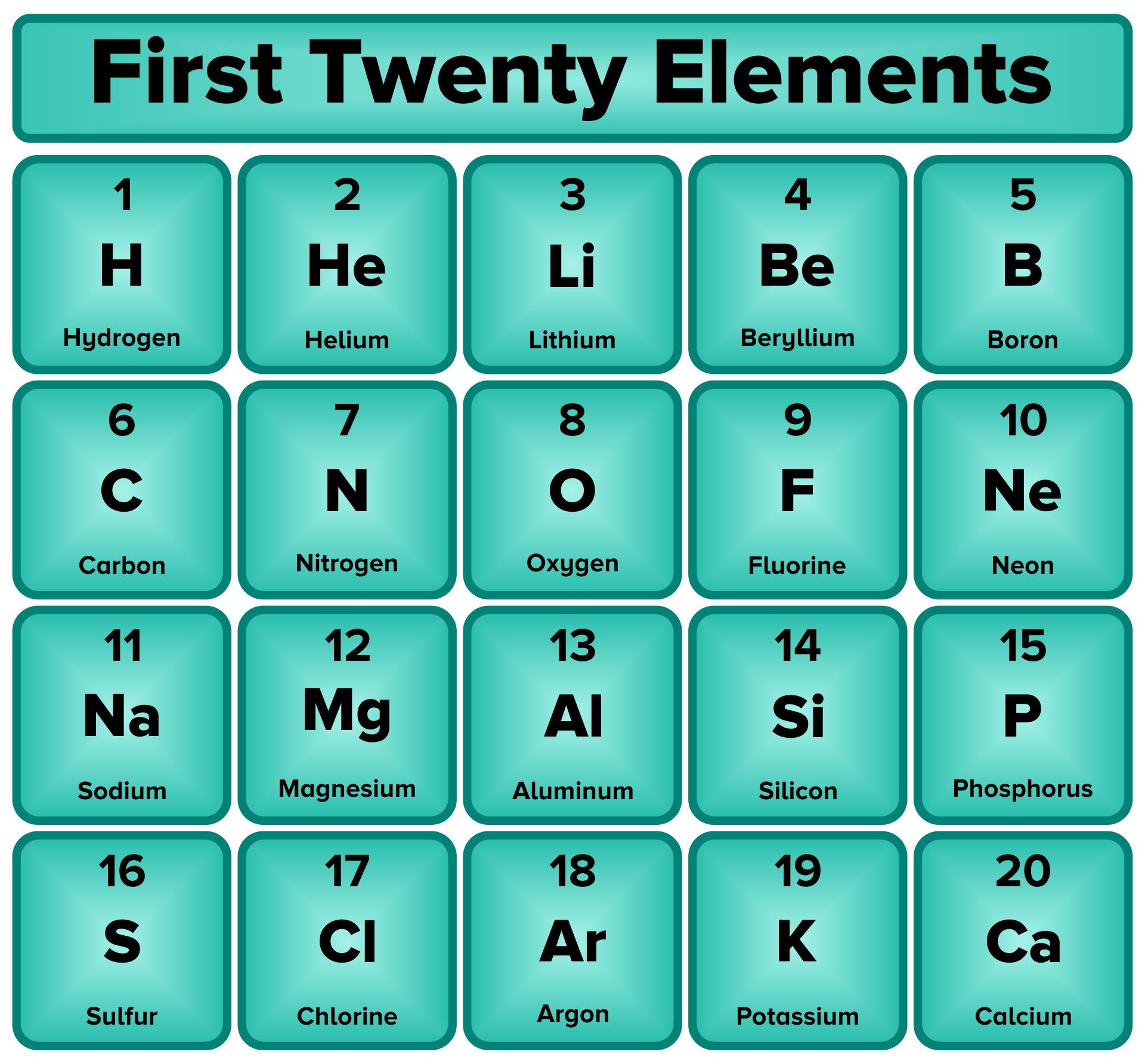
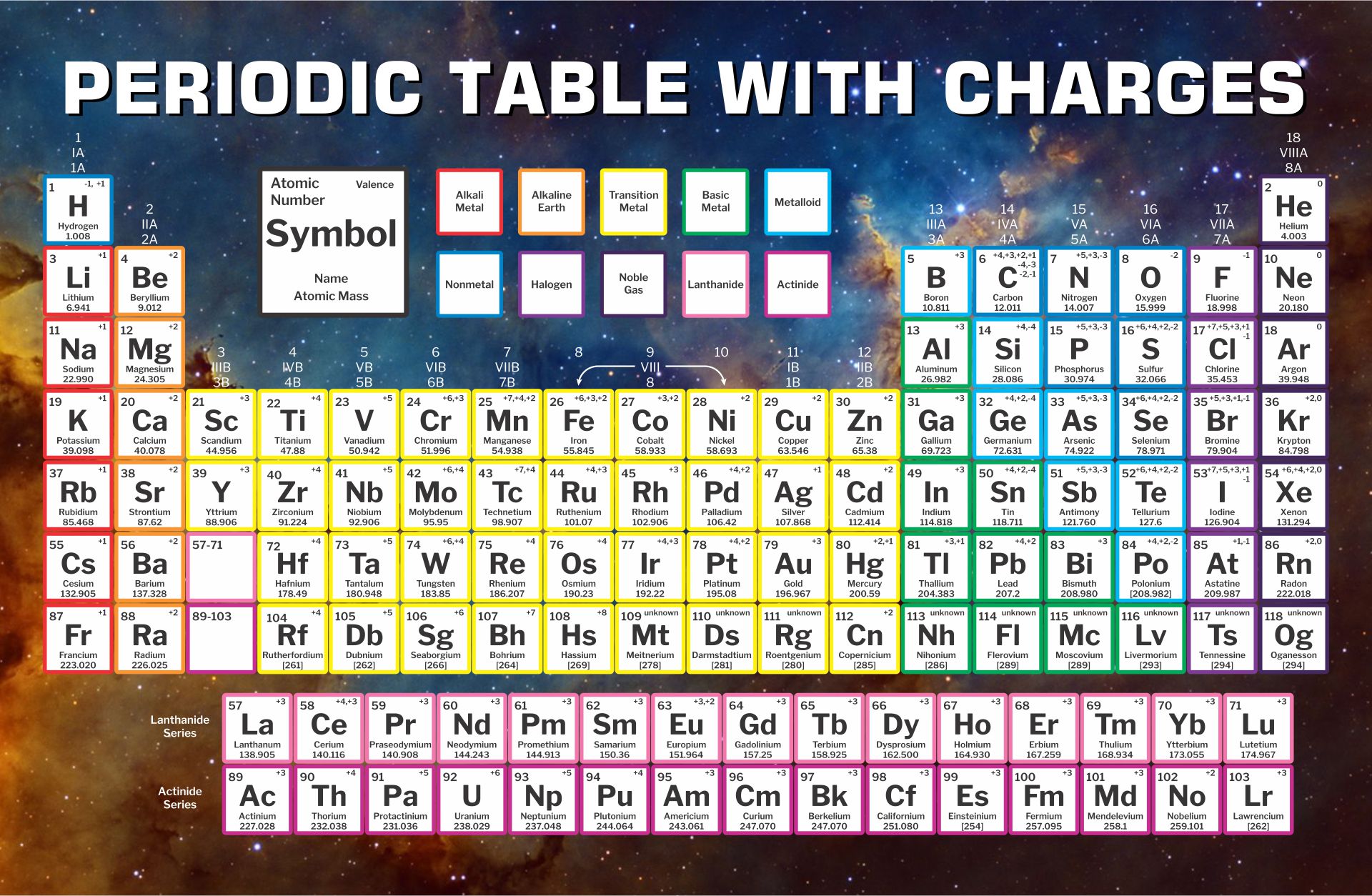
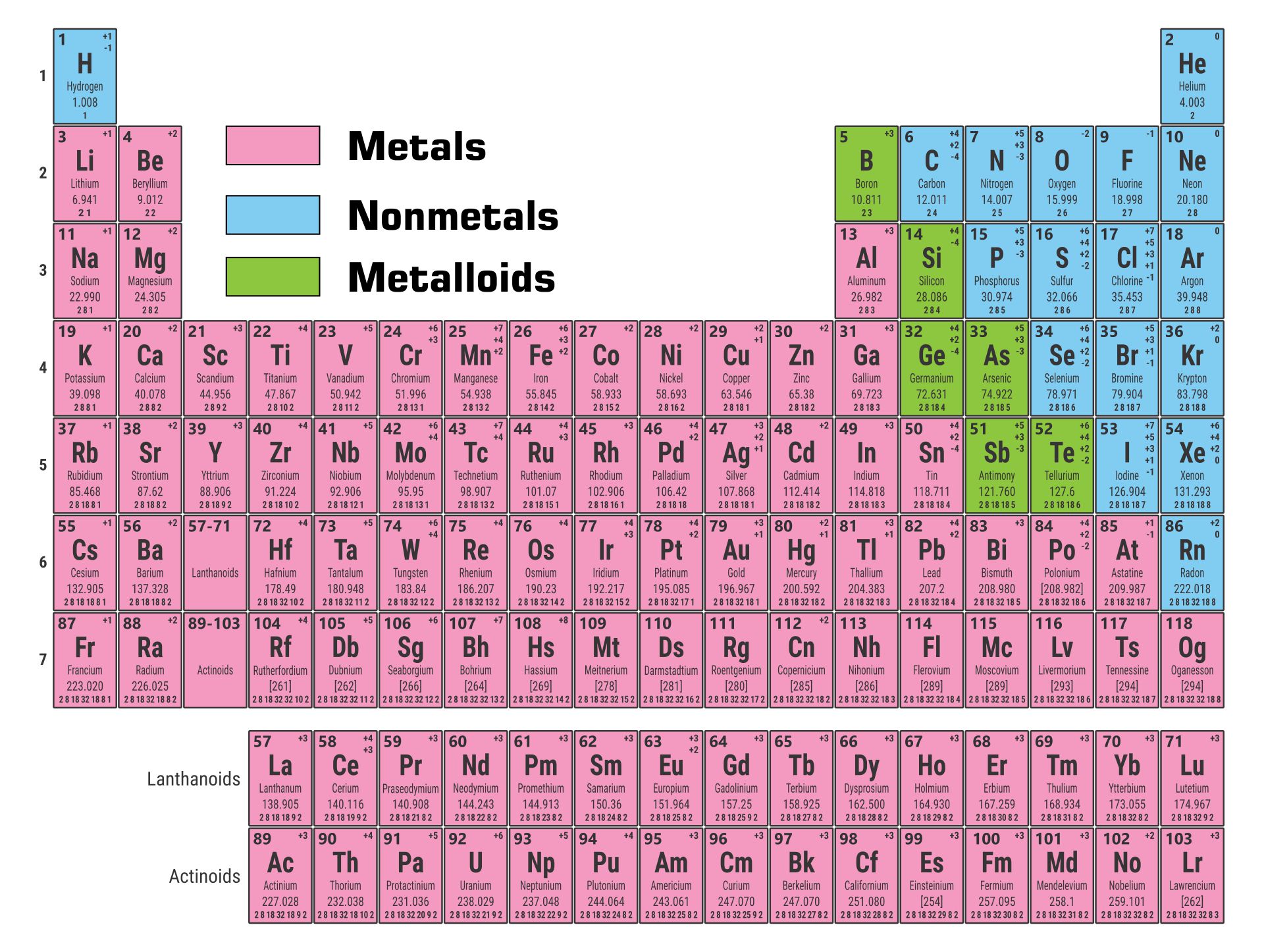
A printable periodic table is a crucial tool for students and educators in the field of chemistry, offering a quick reference to all chemical elements and their properties. It simplifies learning and teaching by providing a visually organized arrangement of elements, helping identify relationships and characteristics at a glance.
Having a large printable periodic table can significantly enhance your study or teaching environment. It serves as an easy-to-view reference for all chemical elements, especially beneficial during group studies, classroom discussions, or when working on projects that require frequent consultation of the periodic table.
A printable periodic table with charges is an invaluable tool for chemistry students, enabling a deeper understanding of ionic compounds and chemical reactions. It outlines the charges of ions, aiding in the quick prediction of compound formation and providing a solid foundation for chemical equation balancing and reaction prediction.
Have something to tell us?
Recent Comments
The printable periodic table resource is a helpful tool for studying and referring to elements in a visually clear and easily accessible format. Great for students and professionals alike!